Wheel of Motion
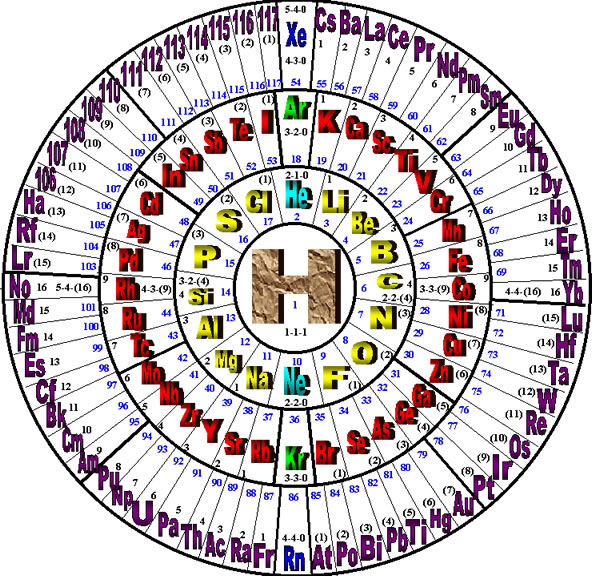
The Wheel of Motion (WoM) representation of the periodic table of elements shows the periodic nature of the elements, as developed in the Reciprocal System of Physical Theory (RST). It was originally developed by Douglas Bundy in 1998, a member of the International Society of Unified Science (ISUS).
Wheel of Motion
The wheel configuration is a more natural way of showing the periodicity of the elements, as they are derived from the Reciprocal System of Theory, than is the traditional table. Each element in the wheel has one additional unit of scalar motion than does its predecessor. Notice that Hydrogen occupies the most prominent position at the center of the wheel, this is appropriate since it is the first (lightest) and most abundant element in the universe. The noble metals occupy the key positions at the top and bottom of each circle. Also, it is immediately apparent from the WoM why the gaps in the standard table appear between the lower numbered rows (rows 1, 2 & 3), while the elements in the highest numbered rows (rows 6 & 7) do not all fit well in the table, and usually have to be shown separately in order to make the size of the table manageable. In the WoM these increasing lengths are naturally accommodated due to the increase in space that each concentric circle provides as each new series (row) begins.
While the RST is a relatively unknown system of physical theory, it predicts that the highest numbered element (heaviest) possible is atomic number 117, which completes the outer circle of the WoM. The atomic numbers of the elements are shown in blue on the inside of the circles, while the three position notation (a-b-c) on the outside indicates the total motion combination of each element in accordance with the RST.
The formula for the total number of elements contained in each circle is 4n2 where n equals the number of the circle beginning with the inner-most or Hydrogen circle. On this basis, the circles have the following number of positions:
The first circle has
- 4(1)2 = 4 positions (three are subatomic & are not shown);
The second has
- 4(2)2 = 16 positions;
The third has
- 4(3)2 = 36 positions;
The fourth has
- 4(4)2 = 64 positions.
Hydrogen’s value is 1-1-1 because it actually occupies the fourth and last position in the center circle, but the three preceding positions are subatomic values and thus are not shown in the WoM. The manner in which the notation proceeds for each succeeding element indicates the total value of its motion and its magnetic and electric configuration in the theory. The first two positions in the notation, (
a-
b-c), are magnetic, or values of two-dimensional scalar motion, while the third position, (a-b-
c), is electric, or values of one-dimensional scalar motion. Hence, n magnitudes in the a or b position are equal to n
2 magnitudes in the c position.
It’s important to note that the RST completely clarifies the existing theoretical picture with respect to the periodic table of elements. Currently, quantum mechanical (QM) calculations, based on the model of the nuclear atom of a nucleus of protons and neutrons surrounded by a cloud of electrons, cannot correctly calculate the periodicity of the atomic order of the elements. The problem is that the Schrodinger wave equation used in this theory requires four quantum numbers to precisely specify the energy levels of the electrons of a given atom, which it then uses to calculate the Coulomb interactions between the electrons and the nucleus. This works fine for the simple atoms, but as the number of electrons grows in the heavier atoms, the equation becomes so complex that it has never been actually solved to this day. As a result, only an approximate calculation is performed that incorporates the first, or principle, quantum number, n. The equation of periodicity derived in this manner is equal to the number of degenerate states for a given level of energy, n (called an energy shell), and is equal to 2n
2, which results in the following periods: The first period has
- 2(1)2 = 2 positions;
The second has
- 2(2)2 = 8 positions;
The third has
- 2(3)2 = 18 positions;
The fourth has
- 2(4)2 = 32 positions.
Comparing this result to the standard periodic table, the first row has 2 elements, hydrogen and helium, and the second row has 8 elements, which both agree with the calculations. However, then things quickly deteriorate from there because the third row also has 8 elements, but the calculation calls for 18 elements. Then, the fourth row of the table has 18 elements, but the calculation shows that it ought to have 32 elements. It’s easy to see what has happened by looking at the Wheel of Motion and comparing it to the table. What is not apparent from the table, is clearly shown by the wheel: the major periodicity of the elements is actually one-half as much as the rows of the table indicate. In other words, it takes two rows of the table to form one period, not one row, and the first period ends with hydrogen, while the second period begins with helium.
Chemists may be reluctant to accept this because it would seem to disrupt the grouping of elements with similar properties in the columns of the table as well as the rows, but this only occurs with the table format. The wheel format can show property groups by their reciprocal positions in the circles, which is more intuitive, if less practical.
However, the more important implication of this result is found in its indictment of QM and the nuclear model of the atom. If there is a much simpler method to make these calculations, and it produces the correct results, which are simple and easy to understand, where does this leave room for QM and the nuclear model of the atom, which are not able to produce correct results?
